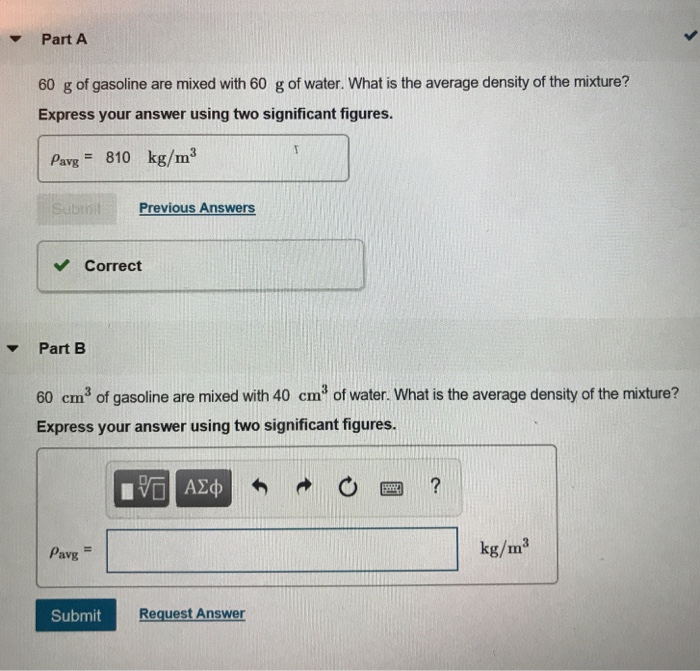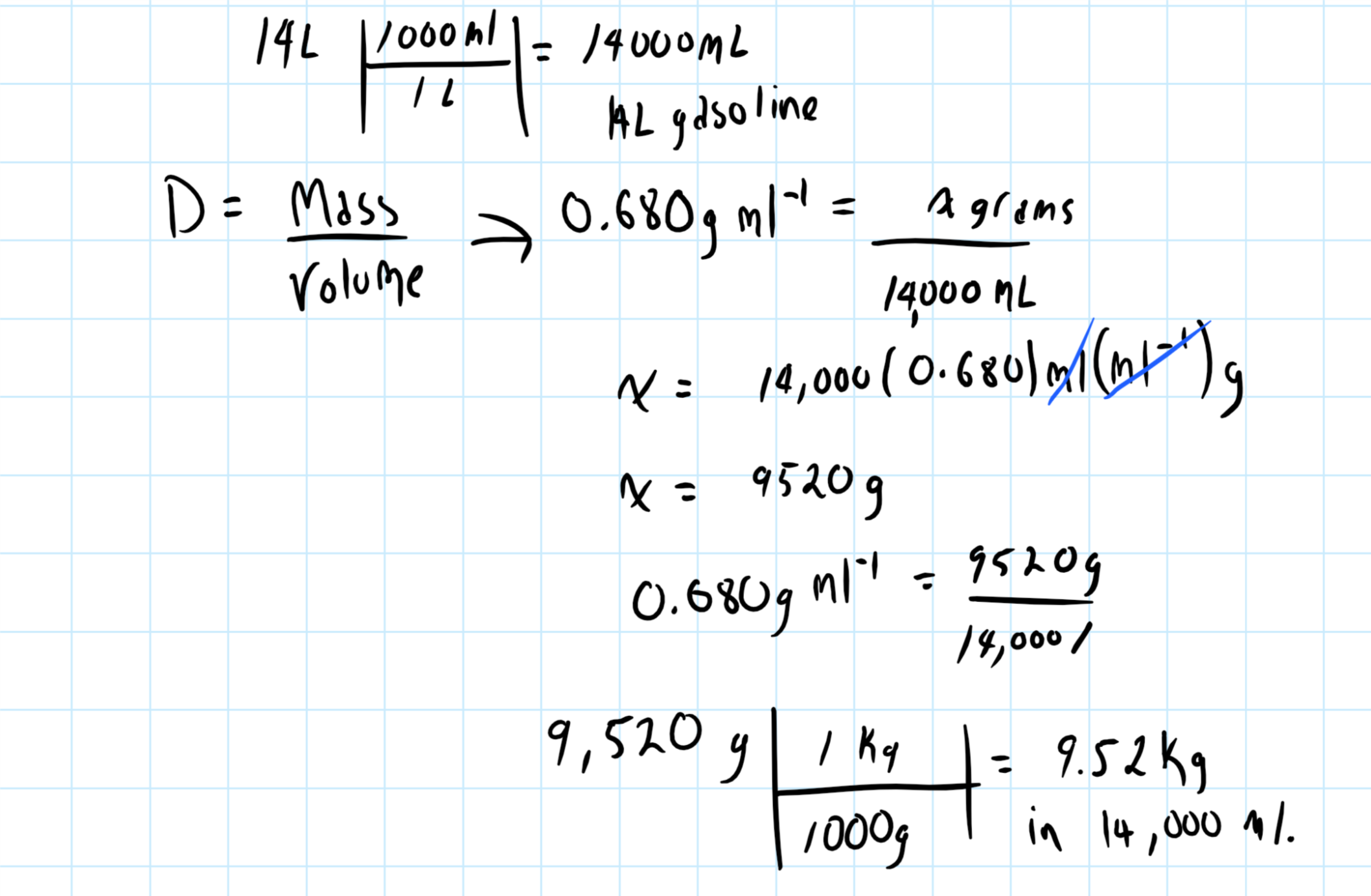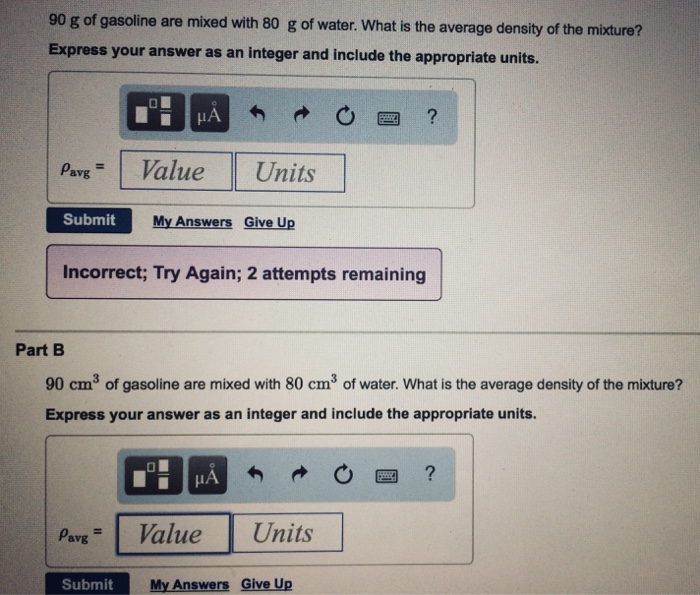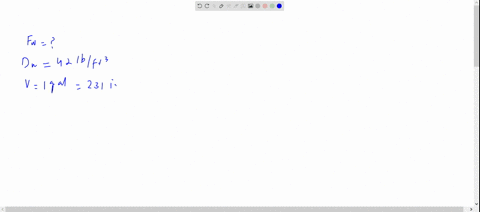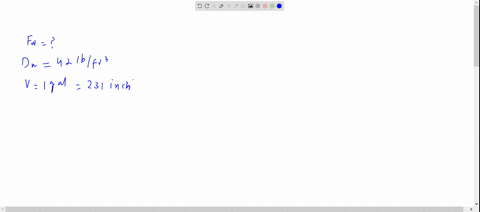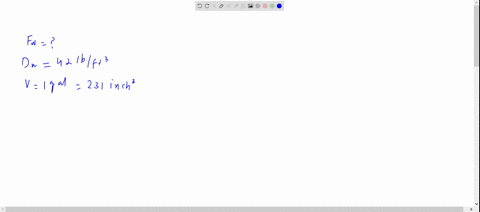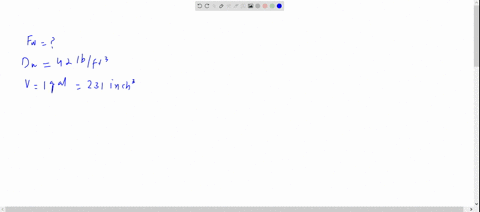
SOLVED: The density of gasoline is 0.774 g/cm3. How much does 20.0 gallons of gasoline weigh, in kilograms?
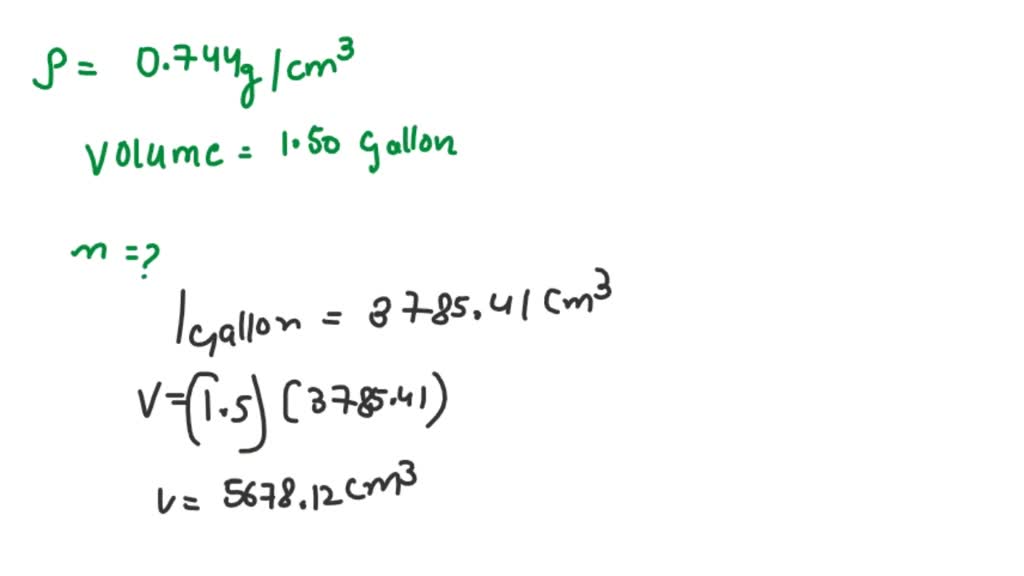
SOLVED: The density of gasoline is 0.774 g/cm3. How much does 1.50 gallons of gasoline weigh, in kilograms?
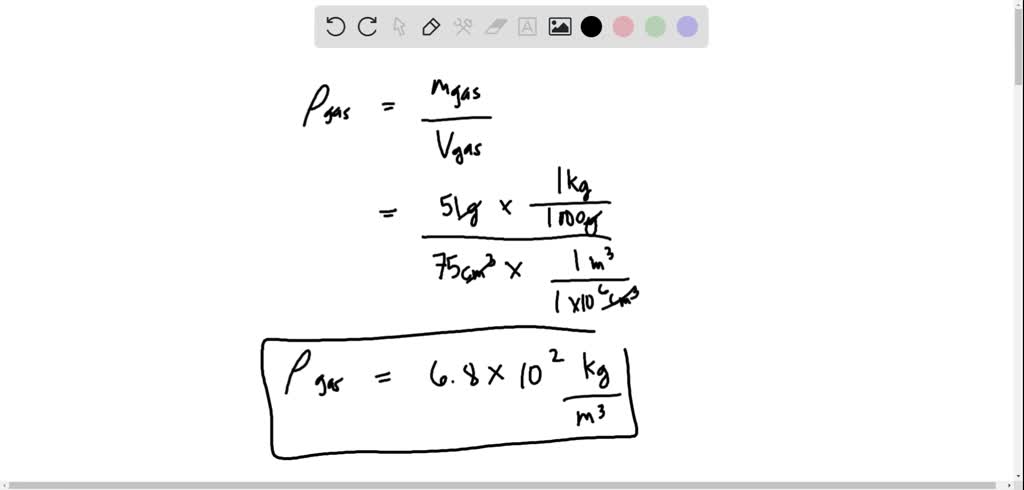
SOLVED:Find the density and specific gravity of gasoline if 51 g occupies 75 cm^3. Make sure you know how to convert cubic centimeters to cubic meters: 1.0 m^3=1.0 ×10^6 cm^3. 1 Density =(

SOLVED: The density of gasoline is 0.774 g/cm3. How much does 20.0 gallons of gasoline weigh, in kilograms?

SOLVED: The density of gasoline is 0.7 g / cm3 and assuming it is made up of octane, C8H18, determine the mass of carbon dioxide (CO2) that is formed when a car

SOLVED: 10. Gasoline has a density of 0.73g/cm'. How many liters of gasoline would be required to increase the mass of an automobile from 1271 grams to 1305 grams?